Boyds has been working with New Deal and a team of European partners on a nanotherapy research project into the treatment of inflammatory bowel diseases.
In Europe, over two million people are affected by ulcerative colitis or
Crohn’s disease, conditions that are characterised by an inflammation of the bowel (Inflammatory bowel diseases). They occur when cells from the immune system become activated and react against contents of the intestine, leaving the patients with fatigue, abdominal pain and severe diarrhoea, blood in stools or even with life-threatening complications such as bowel perforation, bleeding, and in long-standing disease an increased risk of cancer.
The main therapeutic options are conventional drugs or biological therapy. Both inhibit immune cells but increase, as a side effect, the risk of infections. Moreover, they are only effective for limited subsets of patients.
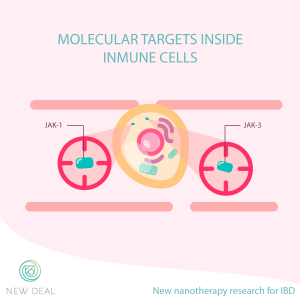 Recent clinical studies testing a new class of drugs have pointed out two potential targets inside immune cells: JAK1 and JAK3. These proteins are needed for immune cell activation, thus, without them, the inflammation will be hampered. Unfortunately, these drugs have a systemic distribution, are not specific, and present adverse-effects.
Recent clinical studies testing a new class of drugs have pointed out two potential targets inside immune cells: JAK1 and JAK3. These proteins are needed for immune cell activation, thus, without them, the inflammation will be hampered. Unfortunately, these drugs have a systemic distribution, are not specific, and present adverse-effects.
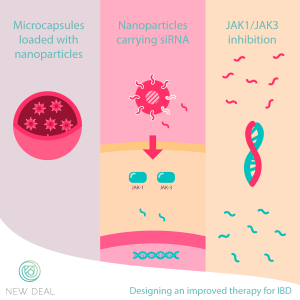
For this reason, New Deal has gathered scientists and companies from all over Europe to join forces and fix this unmet clinical need. The goal is to target JAK1 and JAK3 in a more specific way: using small pieces of genetic material called siRNAs.
These molecules will interfere with the cellular production of JAK1 and JAK3 by binding exclusively the molecules that contain their genetic instructions and impeding the synthesis of these two proteins. Without them, the activation of the cells is hindered, and the inflammation decreases.
But make it into these cells is a challenge. To reach the bowel, the siRNAs will be incorporated into microcapsules; and inside them, lipid nanoparticles will guarantee their stability and allow them to enter the cells. This formulation will allow a local distribution diminishing possible side effects.
With the research project this strategy will be tested in mouse models of the disease in order to provide the Regulatory Agencies a solid preclinical proof-of concept that would open the door to start human clinical trials and test the real capabilities of this therapy in the future.
This video explains the basics about these groundbreaking technologies.
Boyds’ involvement has included:
- Ensuring sufficient information and evidence is generated in the project to support regulatory approval and initiation of a Phase I clinical study
- Compiling an outline clinical development plan providing manufacture, preclinical and clinical design study plans
- Preparation of a briefing package for scientific advice meetings with national regulatory agencies such as the MHRA and the EMA and FDA
- Preparation a CTA/IMPD for a Phase I clinical study
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement number 720905.
 Commenting on the project, Professor Alan Boyd said, “Working with a European team of partners has highlighted the
Commenting on the project, Professor Alan Boyd said, “Working with a European team of partners has highlighted the
importance of sharing expertise and working collaboratively on such an important project.
“With over two million people affected by ulcerative colitis or Crohn’s disease, the incidence of inflammatory bowel disease is increasing, and it is now one of the most common immune-mediated diseases in young adults.
“The Horizon 2020 funded New Deal project aims to develop a highly selective and local therapy with an improved efficacy and safety profile for IBD patients. Using nanoparticles and siRNA New Deal will target JAK1 and JAK3 in a local and specific way to treat inflammatory bowel diseases.”









